|
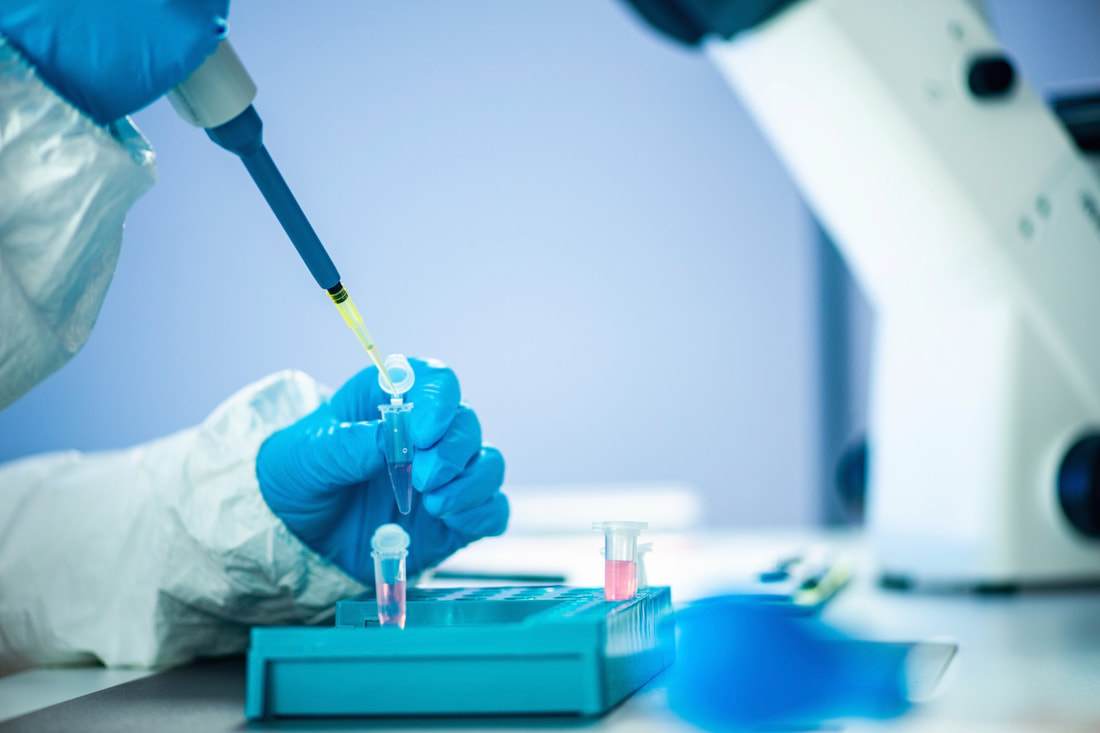
Any clinical and non-laboratory facility you use to conduct safety and toxicology testing in animals may be subject to inspection by FDA or other regulatory bodies. Polaris can prepare you by performing independent audits of sponsor or contract facilities and studies, giving you additional assurance that a lab is in compliance with regulations (e.g., 21 CFR 58) and SOPs.
Polaris performs the following GLP and GCLP Audits:
|
- Home
-
Services
- Good Clinical Practice (GCP) Audits
- Good Laboratory Practice (GLP) and Good Clinical Laboratory Practice (GCLP) Audits
- Good Manufacturing practice (GMP) Audits
- Computer System Validation (CSV)/Part 11 Audits
- CAPA and Remediation
- SOP Development
- Strategic Quality Consulting
- Clinical Project Management (CPM)
- Resources
- About Us
- Contact
|
Over 25 Years of Compliance and Quality Consulting for the Life Science Industry
|
SERVICES
Good Clinical Practice (GCP) Audits Good Laboratory Practice (GLP) Good Clinical Laboratory Practice (GCLP) Audits Good Manufacturing Practice (GMP) Audits Computer System Validation (CSV)/Part 11 Audits CAPA and Remediation SOP Development Strategic Quality Consulting Clinical Project Management (CPM)lljljkkljlkjlkjljk |
© Copyright Polaris Compliance Consultants, Inc.. All Rights Reserved.
Website designed by PROSPER Creative Design